moderna boosters at the FDA
is the romance of "big covid" and federal regulators starting to sour?
moderna is looking to get “booster shots” for their covid vaccine approved by the FDA.
so far, it’s not going terribly well. medpage today (a wonky and non partisan medical rag) was not pulling punches. (HERE) “meh” is a technical term of art in medicine, but i suspect you get the drift.
this “trial” was poorly designed, poorly run, rigged in 10 different ways to show efficacy, and it STILL DIDN’T WORK.
it’s full of lots of nasty little tricks.
let’s take it from the top:
first, this is a biomarker study. it measured antibody levels not a clinical outcome. so that’s a bit of a reach right from the start. it’s an assumptive correlation and one that has not been defined, merely suggested.
second, the inclusion criteria were rigged. they skewed young and healthy vs even the comparator group used to judge antibody levels which was, itself, already skewed young and healthy.
“Overall, 149 adults received the two-dose 100 μg series, which is the dose authorized under EUA, plus the booster dose and were evaluated for immunogenicity. They were a mean age of 53, about three-quarters were between ages 18-65, and 60% were women. Nearly all (95%) were white, and 14% had obesity. Median interval between completion of the primary series and the booster dose was about 7 months.”
these are exactly the low risk folks who will have fewer bad reactions and stronger immune (antibody) reactions.
all the actual high risk folks to whom such boosters would be preferentially offered and whose needs such boosters are purported to serve were excluded.
“FDA staff noted adults with a history of chronic cardiovascular disease, chronic pulmonary disease, HIV, diabetes, and a history of hypertension were excluded from participating in study P201.”
this is like running a drug trial for an obesity drug in only triathletes.
it gets worse.
the rate of covid infection in the trial population was MASSIVE.
this, of course IS a clinical outcome. and whoo doggie does it not align with the assumptive claims of higher antibodies being protective.
“Interestingly, there were 38 COVID-19 cases of 171 booster recipients reported during the Delta surge in study P201.”
guys, that’s a 22% infection rate. i don’t know what population baseline was, but have you seen ANYPLACE where 22% in a fairly healthy group of folks was common? i have not.
this looks terrible.
it looks like our old friend the “immunosuppression worry window” rearing its head.
they they play a scummy trick to try to hide it:
“Moderna performed a post hoc analysis of incidence of SARS-CoV-2 among participants originally randomized to Moderna vaccine (median 13 months post-dose 2) and the participants randomized to placebo and crossed over to Moderna (median 7.9 months, post-dose 2). The analysis showed 77.1 cases per 1,000 person-years versus 49.0 cases per person-years, respectively. However, this analysis was not independently verified by the FDA.”
there’s a meaningful sleight of hand here. it LOOKS like they are compensating for the difference in time in the cohorts by reporting cases per person year. but that’s only half the issue. what was going on DURING that time is the rest. covid’s seasonal expression seems to be around 3-4 months in most places. where it fell and what it was averaged with matters.
example:
imagine new york city in feb of 2020. i take 2 groups of people. one gets nothing. the other gets red sox hats to wear. (GO SAWX!)
we follow the bareheaded group for 8 months. we follow the stylishly hatted group for 13. then we average covid per person year. no hats gets the surge and not much low period to average with. hats get the same but an extra 5 months of low covid time to average in.
“look, red sox hats stop covid!” (told ya, new york)
this is a crazy stunt to try to pull with the FDA and deeply dishonest data handling. it is not normalized at all (nor can it be given temporal divergence). you need to match the period and region so you match the prevalence. everything else is assumptive or hand waving.
ugh.
there’s more.
despite using a “half dose” of 50μg vs 100 for dose 1 and 2:
“FDA staff noted a higher rate of lymphadenopathy among adults ages 18-64 after the booster dose versus the primary series (24.8% vs 11.6%, respectively), but no evidence of "increased frequency or severity of local or systemic reactions after the booster dose, they said. Rates of lymphadenopathy were also significantly higher among adults ages 18-64 than adults ages 65 and up (24.8% vs 5.3%). Fatigue and headache were the most common unsolicited adverse events (AEs)."
so you get the same level of adverse events as before despite half the dose. you get FAR more folks with lymphadenopathy (swollen lymph nodes) a sign of immune activation/stress which by itself makes me wonder about this “no additional AE’s".”
this led me to peruse (and i’m still working on it) the 66 page data submission to the FDA:
a fair few seem to be arguing about this “acceptable safety profile.”
many nordics have suspended moderna use among the young and healthy.

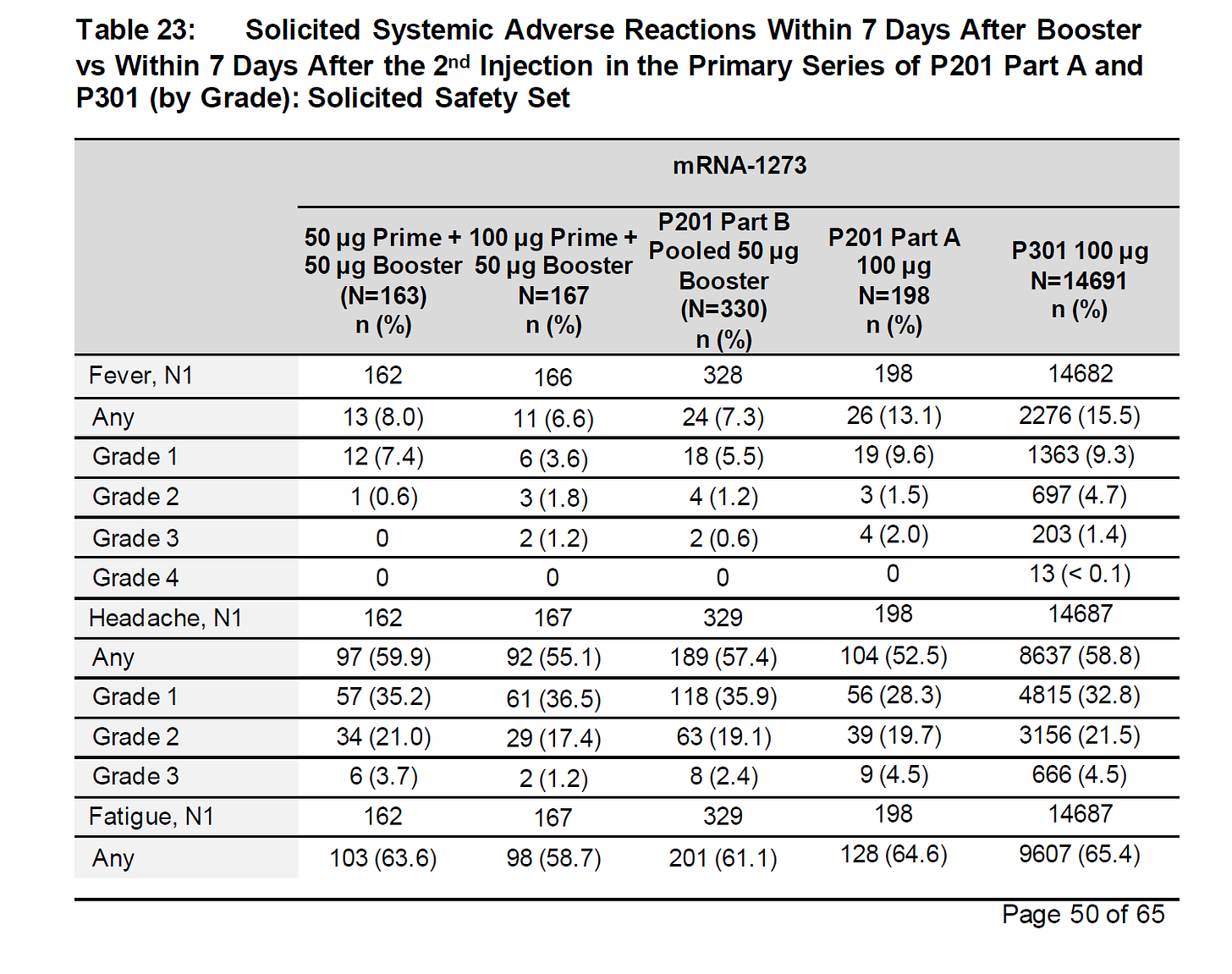
this trial was MUCH too small to catch AE’s like this.
but it still captured many.
~60% got headaches and fatigue.
about half got muscle and joint aches. 15% got nausea. nearly 40% got chills.
this is a lot of people feeling crappy. but, as ever, the real fun is in the UN-solicited AE’s, not the ones they asked everyone about.
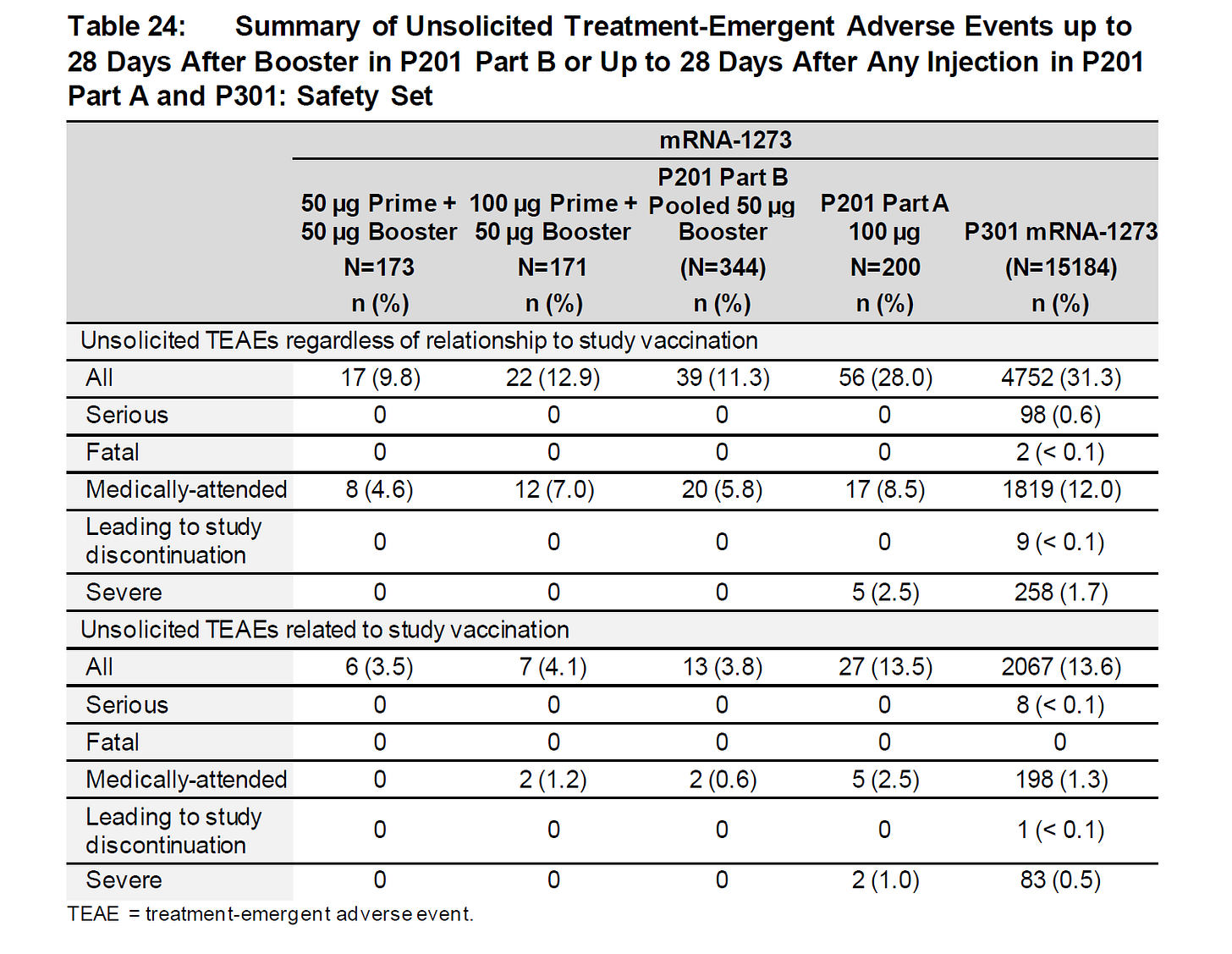
28% of those getting the full 100μg needed medical attention. well no wonder they dropped dose to 50… (also note that it’s 50 that claimed parity to prior, so 100 must have been considerably worse, raising issues about interaction.)
but even then, it’s over 10%. that’s worse than covid in a healthy group like this.
they claim most were “not related to treatment” but this has been a huge fudge in all these studies and i simply do not believe them. the variance is too wide AND it scales heavily with dosage in both arms.
if it’s not related to vaccination, why does a higher dose of vaccination affect it so much?
pull my other paw. it plays jingle bells.
a key thought: if this vaccine is broadly immunosuppressive for a period, whether through TLR deactivation or neutropenia or whatever, you could get all kinds of infections and diseases that look unrelated but are really the result of you being left wide open to infection. if you get an autoimmune response, this too looks unrelated. mRNA vaccines are notorious for triggering autoimmune issues in animals.
it looks to me like this is RJR claiming you cannot prove that cancer was from smoking and defining AE’s out of existence because they are second order effects. (caused by immune suppression or over-activation, not the actual vaccine)
so this trial looked rigged to show efficacy and hide adverse events.
and the capper is: they STILL missed the endpoint.
they claim this:
but the FDA is saying this:
“While "immunobridging analyses against the D614G strain met the pre-specified success criteria for the [geometric mean titers] ratio," FDA staff added that in seroresponse rates after the booster dose compared to dose 2, "the pre-specified success criterion was not met," as the difference in seroresponse rate was -10.5%, and the lower limit of the 95% CI had a criteria of less than -10%”
and, amazingly, seeming to remember that you must specify end points BEFORE a trial and then hit them, not fish around for ones that made it look like your drug worked on a post facto basis and p-hack your way to approval.
it’s a simple system. you say “this is what success looks like. this is how we will test it.” the FDA signs off on your protocol (study design) and your outcomes (endpoints) you file the trial on clintrials and then you do the study.
success state is pre-defined. and the FDA is supposed to hold you to it (and does in most cases).
goodness, might they start actually doing their jobs and running a square game again?
we need to be pro science and pro data, not pro “do something because i’m panicked” and “pharma as talismanic wubbie.”
assessing the data on vaccine does not make you anti-vaxx any more than assessing the data on a bad chemotherapy trail makes you anti-medicine or pro cancer.
it’s, quite literally, what science is. arguing “vaccines are good, this is a vaccine, ergo, it’s good” is just rhetoric by false equivalence.
LOTS of vaccines fail clinical trials. that’s a GOOD thing. it’s why most are so safe.
the FDA was once a gold standard kind of organization. perhaps they are starting to remember themselves.
all in all, this romance of big covid and federal regulators looks to be a bit on the rocks.
it’s long past time.
this has not been working.












The discussion between Joe Rogan and Alex Berenson was interesting when they spoke about how those who got the jabs were behaving. Joe explained (paraphrasing), that this was the first time in history where well educated and wealthy people lined up first to take an experimental drug. Instead of even beginning to admit they might have made a mistake, they have turned angry and now everyone has to get the jabs.
My fear was that the regulatory agencies were likewise brainwashed and would continue to green light these shots. This story from Gato gives me hope that people are reckoning that the numbers do not add up.
The damage that has been done to our society on so many levels is catastrophic, not the least of which is our diminishing trust in government, institutions, and physicians. I’m already considering skipping the flu shot and my yearly physical. Illogical? Not trusting people who lie to me is logical.